There are pros and cons for each type.. Prepare and submit the background package according to above guidance.. It's best to have a draft of the background ...
Feb 2, 2021 — This paper will discuss the scope and procedure for each meeting type, based on FDA's December 2017 Draft Guidance: Formal Meetings ...
6 hours ago — A summary of the meeting's outcome is outlined below.. BFPC instruments are a type of rapid microbiological method (RMM).. They are an ...
Oct 20, 2020 — Instead it'll be about the FDA's approval standards, additional studies the FDA might require of vaccine makers and what type of follow-up should ...
Apr 20, 2021 — However, for Type B meetings, the FDA has up to 21 calendar days from receipt of the meeting request to respond, and if the meeting is granted, ...
14 hours ago — Also, importantly, when drugs receive this type of fast-track approval, ... If those studies fail “to verify clinical benefit, the FDA may” — may ...
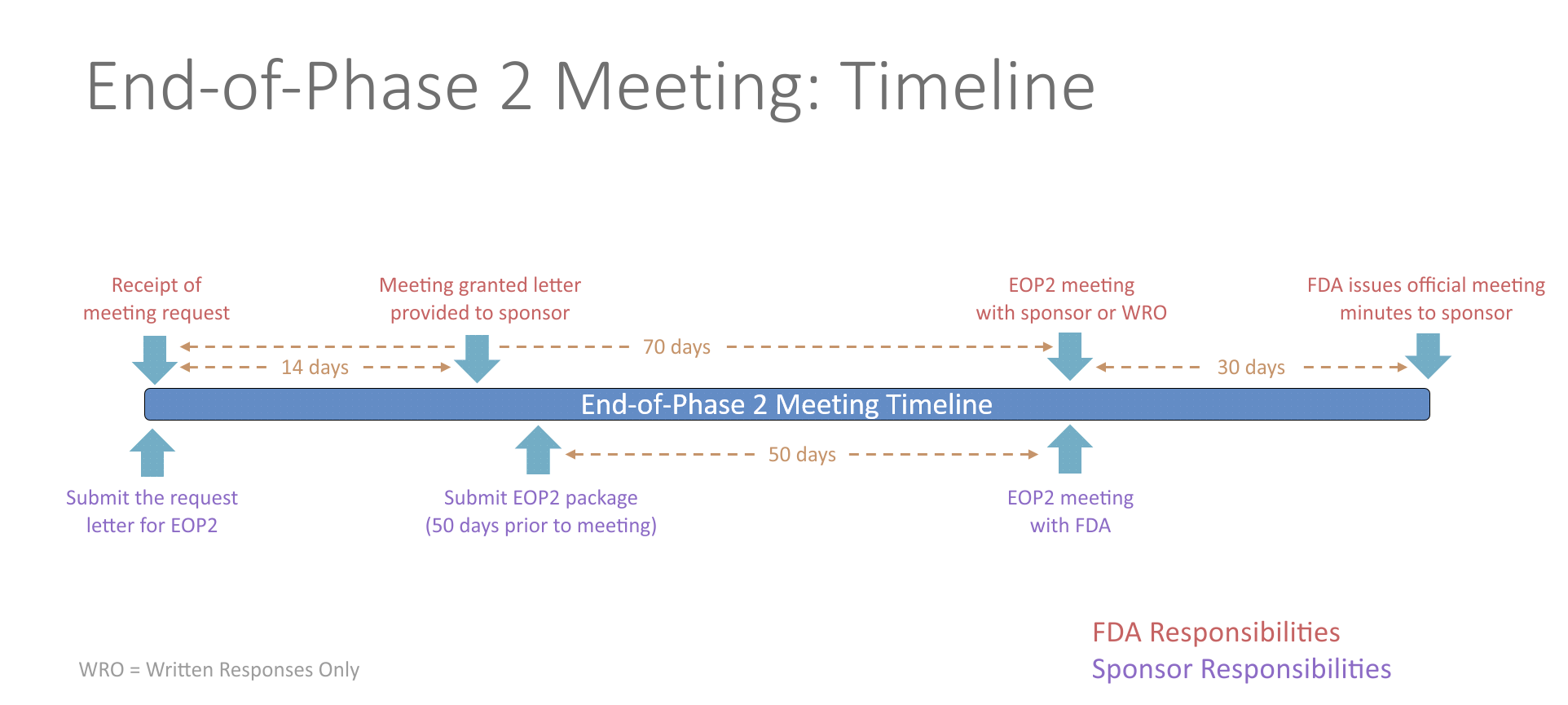
Type B meetings—Most procedural ! meetings such as the pre-IND, EOP2 and pre–NDA meetings fall into this category.. This is the most common ...
Type A meetings are held for development programs that have been stalled or to address an important safety issue, making them critical encounters for sponsors.
A Special Protocol Assessment (SPA) is an advanced declaration from the Food and Drug ... Three types of protocols related to PDUFA products are eligible for this special protocol assessment under the PDUFA goals: ... had been the subject of discussion at an end-of-phase 2/pre-phase 3 meeting with the review division, ...
4 days ago — FDA head calls for inquiry into Alzheimer's drug review ... The meeting came after Biogen stopped two studies because the drug seemed ... “I think the Office of Inspector General is well-equipped to perform this type of work.”.
4 hours ago — The meeting came after Biogen pulled Aduhelm after trials showed it might not have much of an effect.. FDA interactions with drug industry staff ...
TYPE A Critical Path meeting.. Planning the meeting.
DO. https://mmmuhetdin7745.wixsite.com/visthetasu/post/marquette-vs-ucla-live-stream-fbstreams
type meeting
Be polite and prepared.. Bring scientists and data.. Be clear and truthful.. Understand FDA.. Language.Apr 13, 2021 — In these cases, a type of blood clot called cerebral venous sinus thrombosis ... CDC will convene a meeting of the Advisory Committee on ...
Jul 15, 2019 — Mereo BioPharma Group plc. https://magdalenadunn82.wixsite.com/cairehoga/post/memoir-44-air-pack-pdf-download
type meeting minutes
("Mereo").. Mereo BioPharma Announces Successful Type B Meeting with U.S.. FDA and Outlines Accelerated ...
Inspector arrival.. When the inspector arrives, s/he should check in at a specified location.. · Opening Meeting.. The inspector should present his/her credentials to the ...
Oct 28, 2020 — Tricida to Provide Update from its End-of-Review Type A Meeting with the FDA ... SOUTH SAN FRANCISCO, Calif.. --(BUSINESS WIRE)--Oct.. 28, ...
Oct 27, 2020 — 6 to weigh in on whether the U.S.. Food and Drug Administration (FDA) should approve the drug candidate.. The meeting is particularly important ...
Jan 4, 2021 — The FDA typically responds to a sponsor's request for a Type A meeting, if granted, with a meeting scheduled within 30 days from the receipt of ...
Our services: FDA meetings · Prepare and submit meeting request and packages to FDA for various Type A, Type B (Pre-IND and End-of-Phase Meetings) and ...
The meeting types and goal dates were negotiated under PDUFA and apply to formal meetings between. https://katharina84.wixsite.com/conslawnpaddca/post/play-33-free-online-sports-games-gamespook-it-s-all-about-online-free-games
7e196a1c1b

